The U.S. Food and Drug Administration (FDA) has approved Zoryve (roflumilast) topical foam for treating seborrheic dermatitis, also known as scalp eczema, which can cause a scaly, itchy rash on the scalp and skin.
Arcutis Biotherapeutics, the developer, announced that Zoryve foam is cleared for use in adults and children aged 9 and older, and it will be available in the United States by the end of January.
Patrick Burnett, MD, PhD, the chief medical officer at Arcutis, said in the statement, “There has been a real struggle with disease clearance and treatment adherence due to lack of efficacy, difficulty treating certain body areas, inconvenient treatment regimens, and concerns about safety with long-term use. Zoryve foam is a once-daily, steroid-free topical treatment that can be used anywhere on the body, including hair-bearing areas, with no limitation on duration of use.”
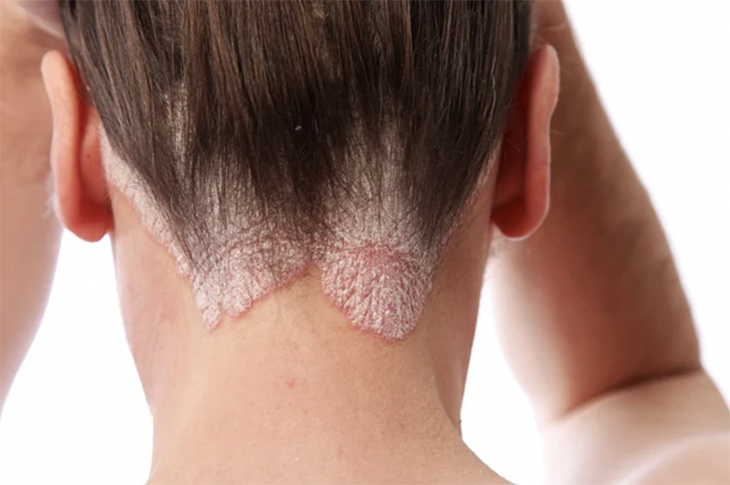
Seborrheic dermatitis is a chronic type of eczema commonly found on the scalp and other areas with oil-producing sebaceous glands. It can cause an itchy rash known as cradle cap in babies and dandruff in adults. More severe cases may lead to thickened skin patches, and persistent scratching can result in serious skin infections.
According to the American Academy of Dermatology (AAD), black people are more susceptible to this condition, and it’s more prevalent in men than in women. The rash may appear pink or purple on darker skin tones and reddish on paler skin tones.
Common Scalp Eczema Symptoms
Common symptoms of scalp eczema include dry, flaky skin, rashes, itching, and burning sensations. It often occurs on the scalp but can also affect the face, eyelids, ears, and areas where skin folds or rubs together, such as the armpits, belly button or skin beneath the breasts.
For mild cases of seborrheic dermatitis, especially those starting on the scalp, treatment typically begins with dandruff shampoo, as recommended by the AAD. However, more severe cases may require the use of topical corticosteroids. It’s important to use corticosteroids for as short a duration as possible due to the risk of skin damage and potential side effects such as high blood pressure, cataracts, and glaucoma associated with long-term use, as advised by the AAD.
A dermatologist at the Dermatology and Laser Center of Chapel Hill in North Carolina, Chris Adigun, MD, says, “Chronic inflammatory conditions have historically been challenging to treat because topical anti-inflammatory steroid creams cannot be used chronically safely.”
“Zoryve is therefore a better option for this chronic condition because it can be used regularly without the risks of chronic topical steroid use, which is most notable for causing thinning of the skin,” Dr. Adigun adds.
How Does Zoryve Work?
Zoryve alleviates inflammation associated with the onset of seborrheic dermatitis. Having previously been approved to treat plaque psoriasis, a condition characterized by skin changes resulting from inflammation, Zoryve demonstrates its efficacy in addressing similar inflammatory processes linked to seborrheic dermatitis.
In a late-stage clinical trial conducted for FDA approval, researchers assigned 304 individuals with seborrheic dermatitis to use Zoryve for eight weeks, while another 153 participants received a placebo foam.
By the study’s conclusion, roughly four out of five patients who received Zoryve reported significant improvement in their skin, with half experiencing complete clearance of their condition.
Moreover, more than 60 percent of Zoryve users also reported substantial reductions in itching, some experiencing relief within just one or two days of beginning treatment, as noted during the 2023 American Academy of Dermatology meeting.
There were very few treatment-related side effects with Zoryve, according to data reported by the meeting. The most common side effects were urinary tract infections, nausea, pain at the application site, and sinusitis.
Danilo C. Del Campo, MD, a dermatologist at the Chicago Skin Clinic, said, “Overall, Zoryve presents several advantages for the treatment of seborrheic dermatitis, including its proven efficacy, user-friendly application, favorable safety profile, and anticipated accessibility.”
“Seborrheic dermatitis, a prevalent and often stubborn dermatological condition that affects a considerable portion of the population, can now be treated more effectively with the approval of Zoryve.”