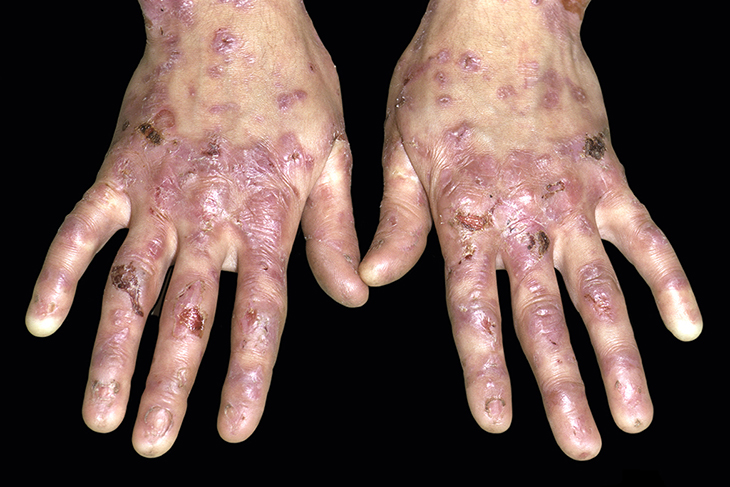
Those who suffer from epidermolysis bullosa (or EB) can tell you how challenging this autoimmune disease can be. They need to be extra careful with everything they do. A simple scratch or a very light touch could affect them in a way that normal people wouldn’t even experience.
They need to live in a bubble because their skin is extremely sensitive. While this disease isn’t quite common, it is debilitating for those who suffer from it. Their only hope at this point is science. They need a cure that would change their lives and make a permanent impact on their wellbeing.
Butterfly Children
Right now, researchers and health experts are looking into a legitimate quality-of-life enhancing treatment that could be available soon for those whose treatments come rarely and efficiently.
Patients who are born with EB are sometimes referred to as “butterfly children.” That’s because they have a disease that prevents their skin cells from coding some types of proteins. What happens is that their skin organ becomes so delicate that even the slightest touch can bring about ruptures and blisters that last for months at a time.
A DNA-coding skin cell is a representation of a genetic therapy option that may be for people who suffer from this sort of disease. When a few drops made were applied onto the wound, they saw a big difference. Under normal circumstances, EB might never heal. However, the latest trial in nine patients, they were able to observe substantial improvements.
What is this cure? It comes in the form of a gel that is known as B-VEC. This is made from the non-replicating shell of a Herpes Type 1 virus that’s been genetically engineered to carry the genetic codes to create a protein called collagen VII. Because they don’t have enough of the strong fibral anchor, which is the component responsible for anchoring different layers of the skin and internal organs together, the result is their inability to produce collagen VII. This is the primary mutation in the DNA that brings about problems for EB sufferers.
They’ve gone through phase 1 and 2 trials on mice. They then shifted it to humans, with the intention for achieving wound surface area reduction, time for wound closure, and duration of wound closure post treatment that came after B-VEC application. The requirements were met. Right now, the parent pharmaceutical company, Krystal Biotech, is looking into the Phase III trials because its efficacy was already proven and seen. They are now trying to get regulatory approval directly to make sure that the miracle gel can be made available for those who need it at the soonest possible time.
Hope for Patients
As for now, they also saw no unfavorable side effects in the trials made. The researchers had nothing but positive report. It’s also a known fact that herpes is extremely difficult to detect, which makes this an excellent vector that can be used for gene therapies because the host immune responses are very rarely set off. While it may seem a little unnatural for scientists to give positive words for Herpes, you should also know that not only is it difficult to detect, but this is one of the only genetic therapy deliver vectors that has the ability to hold the large biological computer file that codes for collagen VII.
The gel made can be applied using a bandage to prevent more damage on the skin. The patients were given the treatment every 1 to 3 days, for 25 days straight. Except for one patient, every single one of them saw that the wounds were healed 3 months after the treatment made. Even better was that the wounds didn’t reopen. When they compared this to the placebo group, the wounds still healed more efficiently and stayed close for longer periods of time.
“It’s not a permanent cure, but it’s a way to really keep on top of the wounds,” study lead and Director of the Blistering Disease Clinic at Stanford Health Care Dr. Peter Marinkovich said in a statement he made. He also said, “It significantly improves patients’ quality of life.”
The results of phase II were published, Markinovich also said about the positive results of second, larger trial of the gel at the 2022 American Academy of Dermatology Annual Meeting in Boston, Mass.
EB does not limit itself to just the skin. In fact, it also affects internal organs that undergo friction. Such examples are the esophagus, corneas, and anus. Markinovich is set to launch a trial to test an endogenous form of the gel that’s meant for treatment of such types of injuries. Krystal, on the other hand, is working to get the FDA approval for the gel.