For patients who have been diagnosed with cancer, chemotherapy is oftentimes the first mode of treatment. While some have fully recovered from cancer with this, there’s no denying that the treatment can take a toll in the body.
Chemotherapy targets both good and bad cells. This is why patients undergoing it are asked to stay home to protect their weak immune system. With just how potent the meds are, scientists have been looking for safe yet alternative forms of treatment.
All about Liquid Biopsy
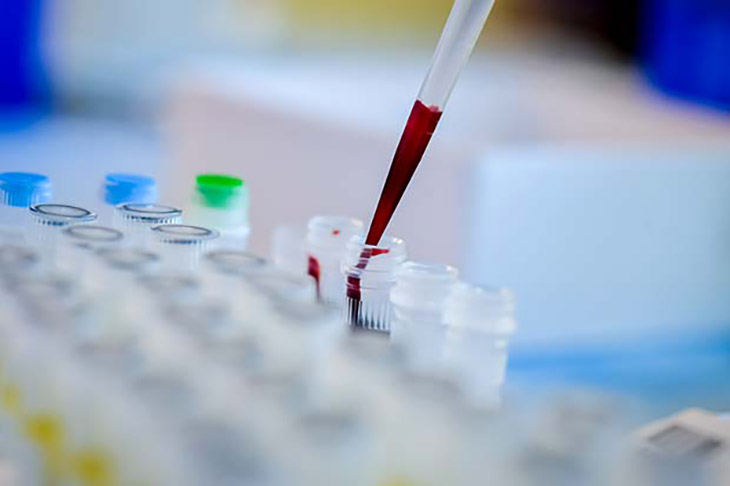
A ‘liquid biopsy,’ or simply a blood test, this may already be able to point out which among the patients diagnosed with stage 2 colorectal cancer need chemotherapy, and who can altogether let go of it. So, what exactly is a liquid biopsy? This blood test is able to screen for fragments of cancer cells found in the bloodstream.
This blood test is able to detect microscopic fragments of genetic materials that are shed when cancer cells die. By doing this, researchers are able to see which patients with stage 2 colon cancer needed supplementary chemotherapy after surgery. They would be able to know who are able to avoid it altogether. This is according to research that was presented last June 4 at the 2022 annual American Society of Clinical Oncology meeting that was held in Chicago.
According to statistics, around 151,030 new cases of colorectal cancer are expected to be diagnosed in the U.S. alone this year. To be able to see which is the most ideal after-surgery strategy for those with stage 2 colon and rectal cancers is a “clinical dilemma,” said lead researcher of the study Jeanne Tie, MD. She is an associate professor at the Walter and Eliza Hall Institute of Medical Research and Peter MacCallum Cancer Center in Victoria, Australia.
Right now, patients are usually given oxaliplatin (Eloxitan). After after surgery, they are then prescribed fluoropyrimidine. The DYNAMIC trial tracked 455 patients in Australia and New Zealand. This is when researchers behind the study utilized a blood test that they call “liquid biopsy.” The method’s main goal is to detect evidence of circulating tumor DNA (ctDNA).
If the patient gets a positive result, this means that the cancer cells are still in their system and that they are still at high risk for recurrence. These individuals have been deemed as good candidates for chemotherapy post surgery. As for patients who tested negative, they were given the option for “watchful waiting.” This merely means that they employ a strategy that requires them to closely watch out for signs of disease recurrence. Or, they may need one round of chemotherapy.
Around 50 percent of the patients who were given a green light to skip chemotherapy based on liquid biopsy findings, decided to forego it. This cut the number of patients down from 28 percent to 15 percent who needed chemo. More importantly, they found no significant difference in disease outcome among those who didn’t opt for more treatment and those who went to get more.
They were all given initially follow-ups every three months and then every six months. The goal was to monitor for possible signs of a relapse. Those who chose “watchful waiting” instead of chemotherapy after they got a negative ctDNA result had recurrence-free survival rates. This was almost identical to those who went in for more treatment.
“Using the blood tests can differentiate the two groups, allowing those without evidence of cancer to forgo additional treatment without relapse risk,” said Dr. Tie.
The study was published in the June 4, 2022, in The New England Journal of Medicine.